AUSTIN -- Abortion opponents on Friday asked a federal court to yank federal approval of a pill used to end pregnancies.
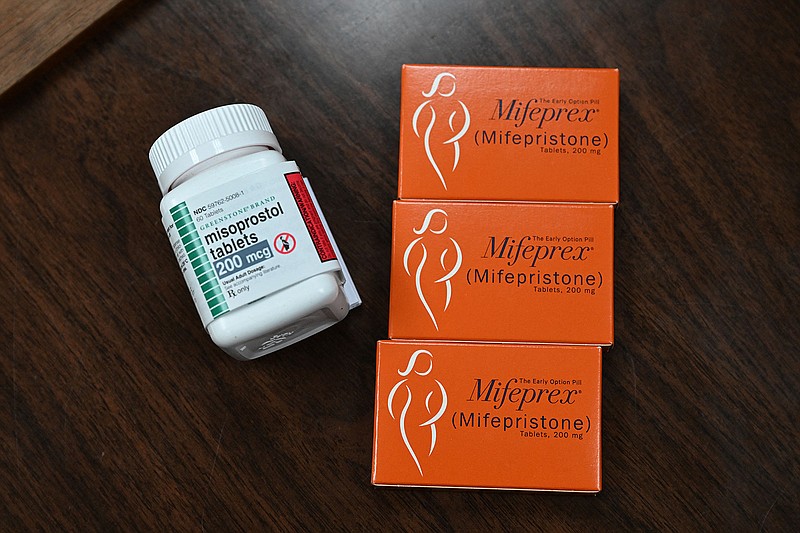
The lawsuit led by the Alliance for Hippocratic Medicine contends the U.S. Food and Drug Administration lacked legal authority to approve and make it easier for people to get the abortion-inducing drug called mifepristone.
The case could have sweeping effects because more than half the abortions in the country are now done with medication. Since the fall of Roe vs. Wade, demand for abortion-inducing pills by mail has surged, especially in states like Texas that have enacted bans.
The FDA said it does not comment on pending litigation.
But in a statement provided to The Wall Street Journal, a spokesperson said the federal agency approved mifepristone "more than 20 years ago based on a thorough and comprehensive review of the scientific evidence presented and determined that it was safe and effective for its indicated use -- medical termination of early pregnancy."
The drug is taken in tandem with misoprostol to block the hormone needed to help a pregnancy develop and to induce contractions. The FDA approved the regimen for use within the first 10 weeks of pregnancy. Research has found medication abortion effective and safe, with serious complications occurring in .3% of patients, according to the Association of American Medical Colleges.
The lawsuit argues the FDA has not sufficiently studied the pills' effect on young women and points to complications some patients experience.
"The FDA has not followed the science, reversed course, or fixed its mistakes -- all to the detriment of women and girls," said the lawsuit.
The Alliance for Hippocratic Medicine, which formed this summer in Amarillo where the suit was filed, includes several religiously affiliated and anti-abortion rights medical groups.
Also among the plaintiffs are four doctors, including one who practices in the Panhandle city of Dumas, and the American College of Pro-Life Obstetricians and Gynecologists. The Alliance Defending Freedom, a conservative Christian legal advocacy group, is representing the group in the lawsuit.
------
Distributed by Tribune Content Agency